ESG – Environmental, Social, and Governance
- Based on our corporate values, we have an organizational culture which appreciates and actively promotes diversity, equity, and inclusion.
- Our research and development process is designed in a way to provide our employees with a safe work environment, to protect the safety of our clinical participants, and to reduce environmental impact.
- We strive to fulfil our social responsibilities through fair and consistent practices and transparent communication with our employees and external stakeholders.
Initiatives

NIT became a participant of the United Nations Global Compact initiative since September 2022. NIT accepts and will take actions to support the Ten Principles of the UN Global Compact and the Sustainable Development Goals (SDGs).
Core Company Values
-
Respect
We strive to always be humble and show respect towards others. This means that we celebrate their successes and empathize with their failures. We appreciate each other’s similarities as well as differences and meet others where they are. Respect is the ruling value.
-
Self-motivation
At the core of our values is hard work and dedication. Through an inner desire to be better, we strive to cultivate and nurture self-motivation in ourselves and in others around us every single day. Positivity begets positivity.
-
Responsibility
Being responsible for our thoughts and actions is at the heart of who we are. We take responsibility seriously. We hold ourselves and our business partners to a very high standard.
Environment l Biosafety
We strive to reduce environmental impact.
-

- By structuring a responsible system for our employees to work from home, we reduce the portion of our carbon footprint caused by commuting.
-

- Complying biosafety management for waste boxes and waste liquid during manufacturing and testing in laboratories.
- Disposing waste & waste liquid with a bio-specialized collector.
-

- Establishing biosafety management guidelines and lab safety policies.
- Providing organizational support to ensure best and safe R&D practices.
- Providing training to R&D staff to ensure sustainable biosafety.
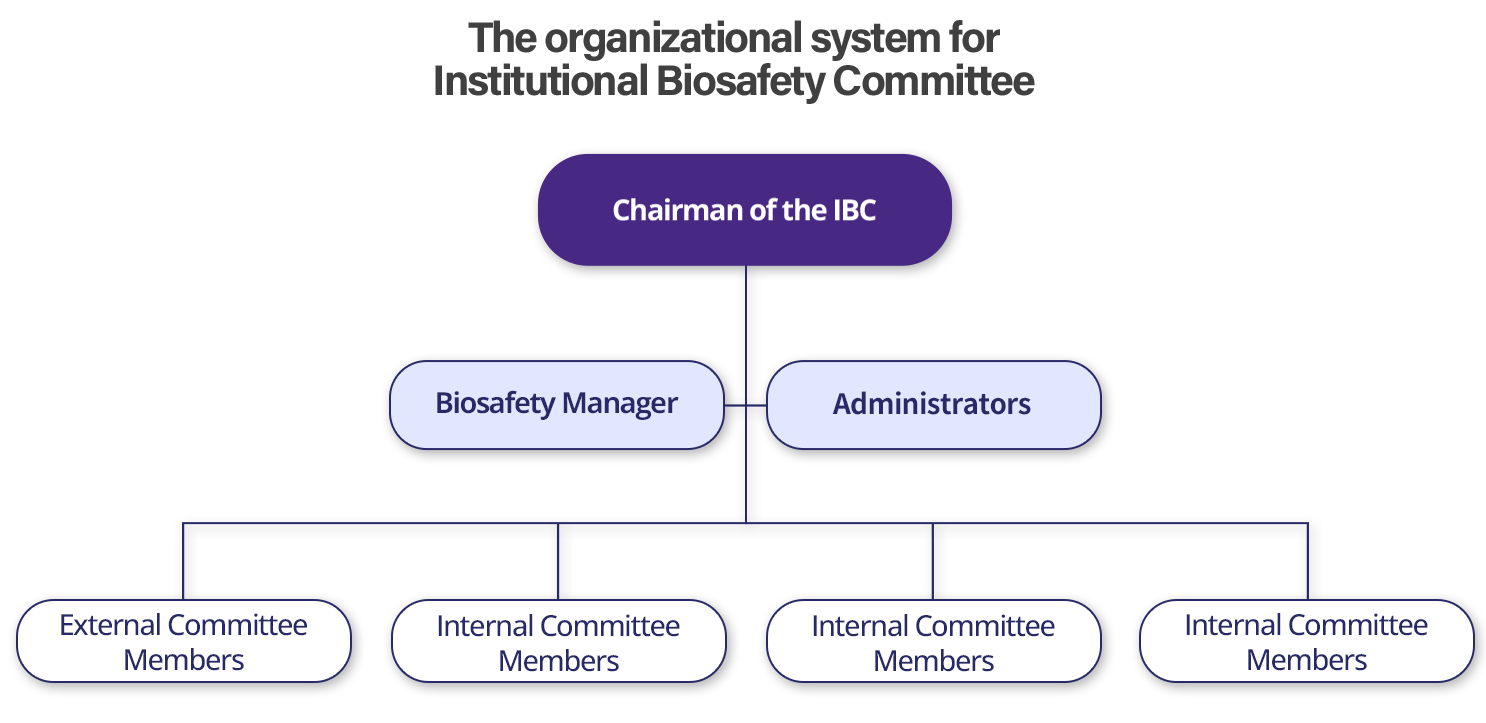
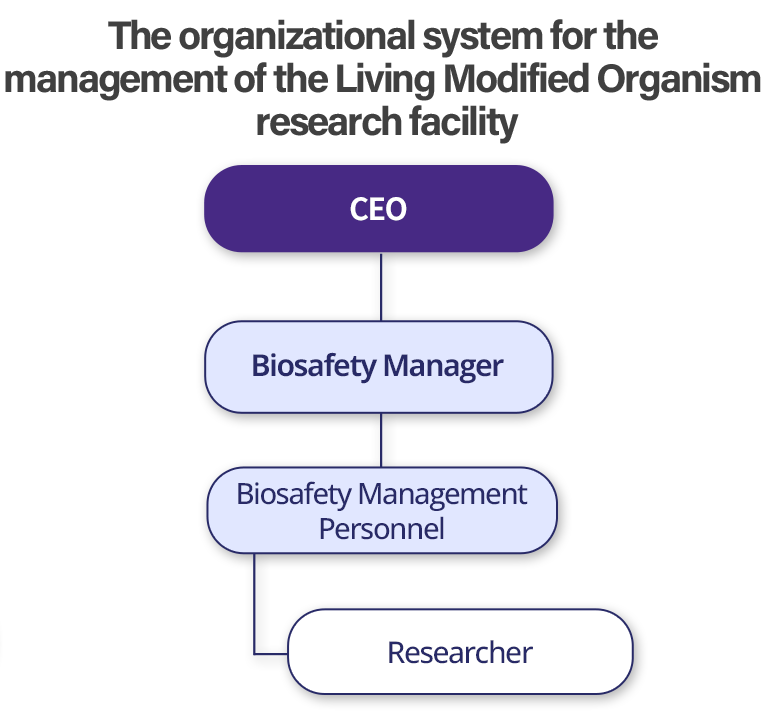
Biosafety Management Structure
Our Employees l
Diversity, Equity, and Inclusion
NeoImmuneTech values diversity, equity, and inclusion and prohibits discrimination on the basis of race, color, religion, age, sex, national origin, disability status, genetics, protected veteran status, sexual orientation, gender identity or expression, or any other characteristic protected by federal, state, or local laws.
We ensure equal employment opportunities(EEO) by establishing policies and procedures that are compliant and follow best practices and interpret policies in a fair and consistent manner. Here are some examples of our proactive approach to promote EEO:
- We select, interview, and hire the most qualified individuals for the positions through fair, consistent, and best talent acquisition practices.
- We prevent harassment and discrimination in the workplace by educating our employees and by having in place a Code of Conduct and an Employee Resolution Program.
- We promote and reward our employees based on objective performance evaluations.
-

Ratio of full-time employees
97.8% -

Ratio of female employees
49.3% -

Racial/ethnic diversity
85%(US office: 69%) -

Ratio of fully remote employees
16%
(As of 2021)
Social Contributions l
Community Relations
NeoImmuneTech is operating the ‘Pohang Innovation Salon (PIS)’ to share Information and experiences about bio and healthcare industry with POSTECH in Pohang, where the research institute is located.
- After the first PIS in May 2021, lectures were held by about 30 experts.
- About 400 industry experts and researchers participated to exchange knowledge about the bio industry and business.
Safety l Laboratory
& Data Security
NeoImmuneTech has established policies and procedures to ensure workplace safety.
- Workplace Safety and Security
- Drug-Free Workplace
- Alcohol-Free Workplace
- Smoke-Free Workplace
We have established information security policies and practices to ensure the security of all sensitive, proprietary, and confidential information.
- General Data Protection Regulation (GDPR) compliance implemented for clinical data safety.
- Mandatory security pledges and mandatory information security programs.
Product Quality l
Global Compliance
NeoImmuneTech, as a biotech company dedicated to development of novel immunotherapeutics, we comply with all applicable laws and regulations for the health and safety of patients.
- Compliance with GCP (Good Clinical Practice) to enhance reliability and ensure patients safety.
- Manufacture according to cGMP/GMP (current Good Manufacturing Practice) management procedures through a contract manufacturing organization (CMO) approved in advance by regulatory authorities such as the FDA and EMA.
Governance
NeoImmuneTech adopted its certificate of incorporation and bylaws—which govern the company’s operations—in accordance with Delaware General Corporation Law, with the aim of establishing a transparent governance structure. NeoImmuneTech holds its board meetings and shareholder meetings pursuant to the certificate of incorporation and the bylaws.
- The board of directors (BOD) consists of five (5) members: one (1) inside director, three (3) outside directors, and one (1) other non-executive director.
Composition of BOD
| Name | Title | R&R | Experiences | Date of appointment |
|---|---|---|---|---|
| Se Hwan Yang | Executive Director (Registered/Full-time) |
President (CEO) |
(Current) NeoImmuneTech, Inc. CEO (Former) Genexine, Inc. Head of Research Institute and Business Division |
2022-03-31~2025-03-31 |
| Jung Won Woo | Non-Executive Director (Registered/Part-time) |
Non-Executive Director | (Current) Genexine, Inc. CEO (Former) Genexine, Inc. President of Research Institute |
2021-03-31~2023-03-31 |
| Soo-Hyoung Kang | Independent Director (Registered/Part-time) |
Independent Director | (Current) Aimed Bio Inc. Advisor (Former) Aimed Bio Inc. CEO |
2022-03-31~2024-03-31 |
| Byoung Jo Kang | Independent Director (Registered/Part-time) |
Independent Director | (Current) B.J Kang Law.PLC CEO (Former) Arlington International Tax Firm Flott & Co. Attorney |
2021-03-31~2024-03-31 |
| Jin-Hyoung Kang | Independent Director (Registered/Part-time) |
Independent Director | (Current) Seoul St.Mary's Hospital, The Catholic University of Korea, Professor (Former) Gangnam St.Mary's Hospital, The Catholic University of Korea, Associate Professor |
2021-03-31~2024-03-31 |
In order to secure the independence of the company’s management, the company has minimized the number of directors designated by the largest stockholder to one (1). Also, transactions between the company and the largest stockholder could be considered as interested transactions under the Delaware General Corporation Law, and as such, the director designated by the largest stockholder is recused with respect to any portion of the board meeting during which any interested transaction is discussed, considered, voted, or adopted. Currently, Genexine, Inc. is the largest stockholder, and Jung Won Woo is the director designated by Genexine, Inc. All other directors, including three outside directors, have no special interest in Genexine, Inc.
BOD activities
| Round | Date | Agenda | Result | Attended/All | Note |
|---|---|---|---|---|---|
| 21-01 | Jan. 04, 21 | 1) Approval of Chief Financial Officer (CFO) bonus | Approved | 6/6 (2/2) |
- |
| 21-02 | Jan. 06, 21 | 1) Issuance of IPO stock and additional stock issuance within a maximum of 5% of IPO stock 2) Conclusion of acquisition contract |
Approved | 6/6 (2/2) |
- |
| 21-03 | Jan. 20, 21 | 1) Approval of conclusion of Chief Operating Officer (COO) employee contract | Approved | 6/6 (2/2) |
- |
| 21-04 | Jan. 29, 21 | 1) Convenience of general stockholders’ meeting and set date of convening general meeting of stockholders | Approved | 6/6 (2/2) |
- |
| 21-05 | Feb. 16, 21 | 1) Approval of appointment of Chief Scientific Officer (CSO) 2) Approval of signing of CFO employment contract 3) Confirmation of joint research contract with YBL |
Approved | 6/6 (2/2) |
- |
| 21-06 | Mar. 08, 21 |
1) Approval of consolidated and separate financial statements for 2020 2) Approval of signing of employment for Chief Executive Officer (CEO) 3) Approval of incentive rules |
Approved | 6/6 (2/2) |
- |
| 21-07 | Mar. 16, 21 |
1) Approval of grant of stock option 2) Approval of the agenda at the regular general meeting of stockholders |
Approved | 6/6 (2/2) |
- |
| 21-08 | May. 27, 21 |
1) Approval of signing of employment contract for Chief Business Officer (CBO) 2) Approval of changes to the operating regulations of the BOD 3) Approval of signing an agreement on the succession of the lessee’s status in relation to the sub-lease contract of the research institute affiliated with the company |
Approved | 5/5 (3/3) |
- |
| 21-09 | Jun. 04, 21 | Approval of cancellation of grant of stock option | Approved | 5/5 (3/3) |
- |
| 21-10 | Jun. 26, 21 | Approval of cancellation of grant of stock option | Approved | 5/5 (3/3) |
- |
| 21-11 | Jul. 26, 21 | Approval of signing a contract area change agreement with Genexine Inc. related to the sub-lending contract of the research institute affiliated with the company | Approved | 5/5 (3/3) |
- |
| 21-12 | Jul. 29, 21 |
1) Approval of cancellation of grant of stock option 2) Changes in stakeholder transaction rules 3) Approval of annual limit on stakeholder transactions 4) Approval of changes to the operating regulations of the board of directors 5) Target bonus payment and Chief Clinical Officer (CCO) appointment according to Dr. Jing (“Jean”) Fan’s one-year evaluation |
Approved | 5/5 (3/3) |
- |
| 21-13 | Aug. 17, 21 | Approval of the Chief Business Officer (CBO) to set goals for 2021-2022 | Approved | 5/5 (3/3) |
- |
| 21-14 | Sep. 28, 21 | 1) Approval of stock option exercise guidelines 2) Approval of stock option grant |
Approved | 5/5 (3/3) |
- |
| 21-15 | Dec. 15, 21 | Establishment of the base date for the 8th regular general meeting of stockholders | Approved | 3/5 (3/3) |
- |
| 21-16 | Dec. 21, 21 |
1) Approval of transfer of address of subsidiaries 2) Approval of the establishment of internal accounting management regulations 3) Approval of loans to executives and employees 4) Approval of establishment of contract approval regulations and signing authority regulations |
Approved | 5/5 (3/3) |
- |
| 22-01 | Feb. 15, 22 |
1) Approval of fiscal year 2021 financial statements and business reports 2) Appointment and delegation of external auditors 3) Convocation of a regular general meeting of stockholders 4) Cancellation of grant of stock options |
Approved | 5/5 (3/3) |
- |
| 22-02 | Feb. 15, 22 | Approval of changes to the COO, CSO, and CFO employment contract | Approved | 5/5 (3/3) |
- |
NeoImmuneTech provides this link as a courtesy and does not control the content of the site you are about to enter.
Do you wish to continue?