SCIENCE
NT-I7 is a Unique T Cell Amplifier.
Interleukin-7 (IL-7) is Central to T Lymphocyte Development and Survival.
IL-7 acts through IL-7 receptor (IL-7R), which is expressed on naïve and memory CD4+ and CD8+ T cells. Thus, IL-7 promotes proliferation, maintenance, and functionality of these key T cell subsets mediating immune responses. On the other hand, IL-7R is expressed on regulatory T cells at low levels, which is a T cell subset involved in limiting T cell responses4.
At NeoImmuneTech, we are exploring the utility of IL-7 based therapeutics in enhancing immune function.
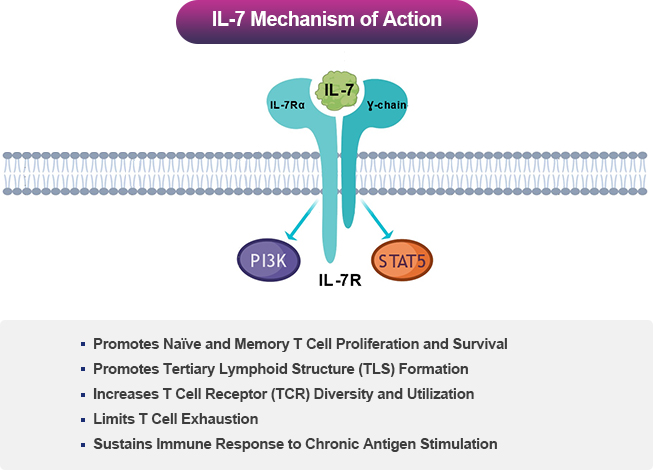
NT-I7 is a More Potent, Stable, and Long-Acting Human IL-7.

NT-I7 Boosts T Cell Numbers and Functionality to Enhance Immune Function and Potentially Provide Greater Therapeutic Benefits to Patients.
Help in Cancer Treatment See How NT-I7 Could
Enhance Immunity to Infectious
Diseases See How NT-I7 Could
Improve Immunological
Disorders See How NIT is Advancing
NT-I7 in Clinical
Development

1. Mazzucchelli, Renata, and Scott K. Durum. "Interleukin-7 receptor expression: intelligent design." Nature Reviews Immunology 7.2 (2007): 144-154.
2. Fry, Terry J., and Crystal L. Mackall. "Interleukin-7: from bench to clinic." Blood, The Journal of the American Society of Hematology 99.11 (2002): 3892-3904.
3. Ponchel, Frederique, et al. "Interleukin-7 deficiency in rheumatoid arthritis: consequences for therapy-induced lymphopenia." Arthritis Res Ther 7.1 (2004): R80.
4. Seddiki, N. Santner-Nanan B, Martinson J, Zaunders J, Sasson S, Landay A, Solomon M, Selby W, Alexander SI, Nanan R, Kelleher A, Fazekas de St Groth B. "Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells." J Exp Med 203 (2006): 1693-1700.
NT-I7 in Oncology
T Cells Play an Important Role in Fighting Cancer.
NeoImmuneTech is conducting clinical trials to evaluate how NT-I7 can bring benefit to cancer patients in different settings, as a monotherapy and in combination with other anti-cancer treatments, including immunotherapies.
NT-I7 Enhances T Cell-Mediated Anti-Tumor Immunity through Multiple Mechanisms.
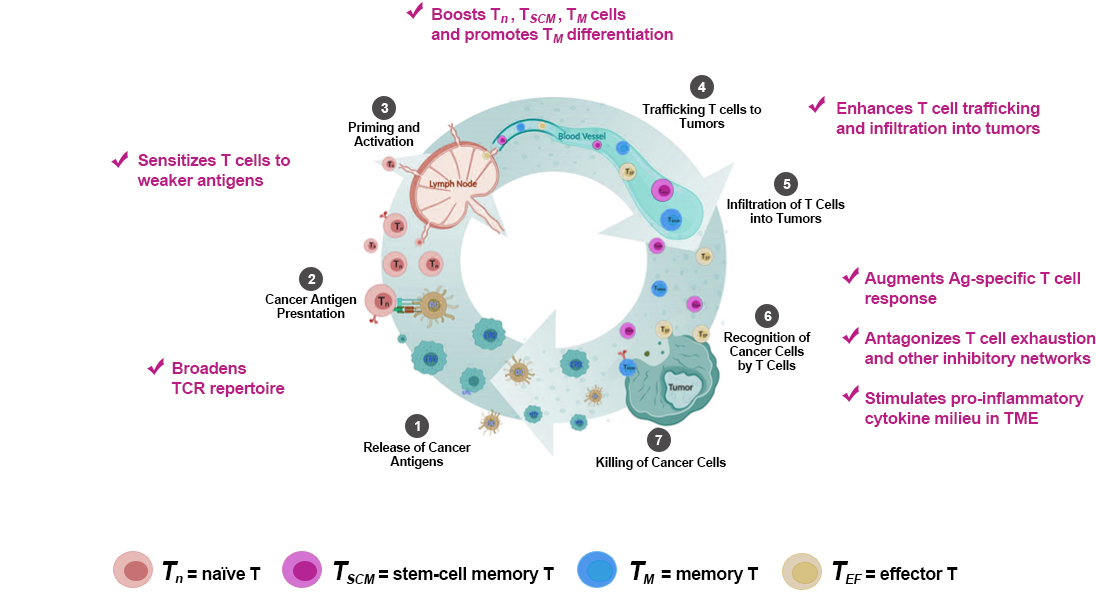
NT-I7 is Uniquely Positioned to Address Unmet Medical Needs in Immuno-Oncology (I-O).

Visit our Scientific Publication section for more information on NT-I7 for Oncology.
NIT is collaborating with major cancer centers and universities in the U.S. and South Korea for our clinical trials with NT-I7. To learn about our on-going cancer clinical trials, please click Here .
NT-I7 in Infectious Diseases
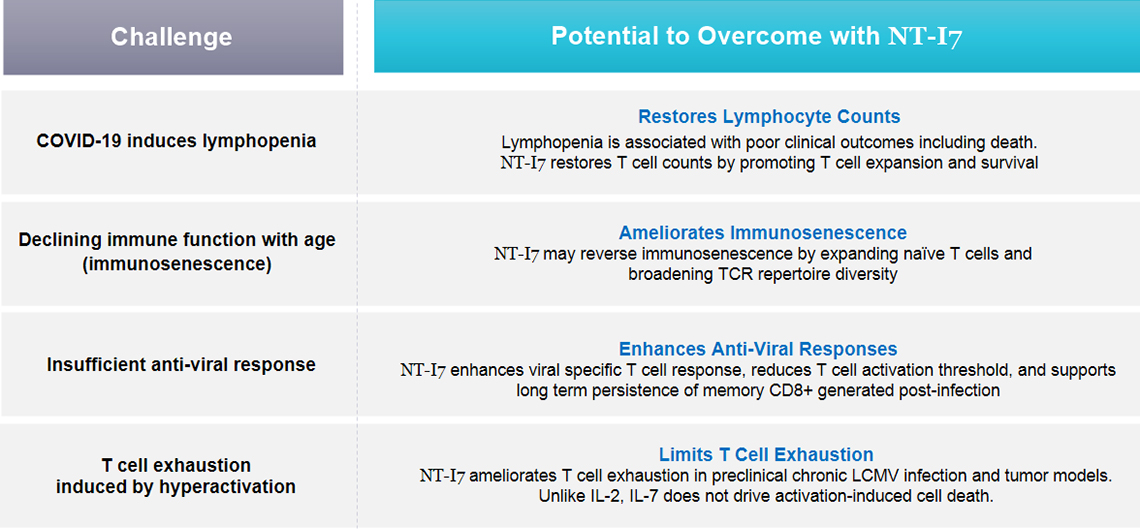
NT-I7 may Improve Immune Response to COVID-19 through Multiple Mechanisms
IL-7 plays a crucial role in T cell homeostasis and can induce rapid expansion of multiple T cell populations, while also preventing apoptosis and exhaustion of effector T cells. Thus, exposing T cells to NT-I7 may expand T cell counts, prevent T cell exhaustion, enhance T cell effector functions, and thereby improve clinical outcomes for COVID-19 patients.
In clinical trials, NT-I7 has demonstrated the ability to increase absolute lymphocyte counts, including naïve and memory CD4+ and CD8+ T cells, and exhibited a well-tolerated safety profile.
NT-I7 has the Potential to Enhance Vaccines
1. Guan W, Ni Z, Hu Y et al. “Clinical Characteristics of Coronavirus Disease 2019 in China.” New England Journal of Medicine. 2020;382(18):1708-1720.
2. Huang C, Wang Y, Li X et al. “Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China.” The Lancet. 2020;395(10223):497-506.
3. Tan L, Wang Q, Zhang D et al. “Lymphopenia predicts disease severity of COVID-19: a descriptive and predictive study.” Signal Transduct Target Ther. 2020;5(1).
4. Terrazzini N, Mantegani P, Kern F, Fortis C, Mondino A, Caserta S. “Interleukin-7 Unveils Pathogen-Specific T Cells by Enhancing Antigen-Recall Responses.” J Infect Dis. 2018;217(12):1997-2007.
5. Deshpande P, Cavanagh M, Le Saux S, Singh K, Weyand C, Goronzy J. “IL-7– and IL-15–Mediated TCR Sensitization Enables T Cell Responses to Self-Antigens.” The Journal of Immunology. 2013;190(4):1416-1423.
6. Mackall C, Fry T, Gress R. “Harnessing the biology of IL-7 for therapeutic application.” Nature Reviews Immunology. 2011;11(5):330-342.
7. Zheng M, Gao Y, Wang G et al. “Functional exhaustion of antiviral lymphocytes in COVID-19 patients.” Cell Mol Immunol. 2020;17(5):533-535.
8. Zheng H, Zhang M, Yang C et al. “Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients.” Cell Mol Immunol. 2020;17(5):541-543.
9. Yang X, Dai T, Zhou X et al. “Analysis of adaptive immune cell populations and phenotypes in the patients infected by SARS-CoV-2.” 2020.
NIT is collaborating with major cancer centers and universities for our clinical trials with NT-I7. To learn about our on-going clinical trials in infectious diseases, please click Here .
NT-I7 in Immunological Disorders
T Cells are Essential to the Immune System’s Defense Against Foreign Pathogens.
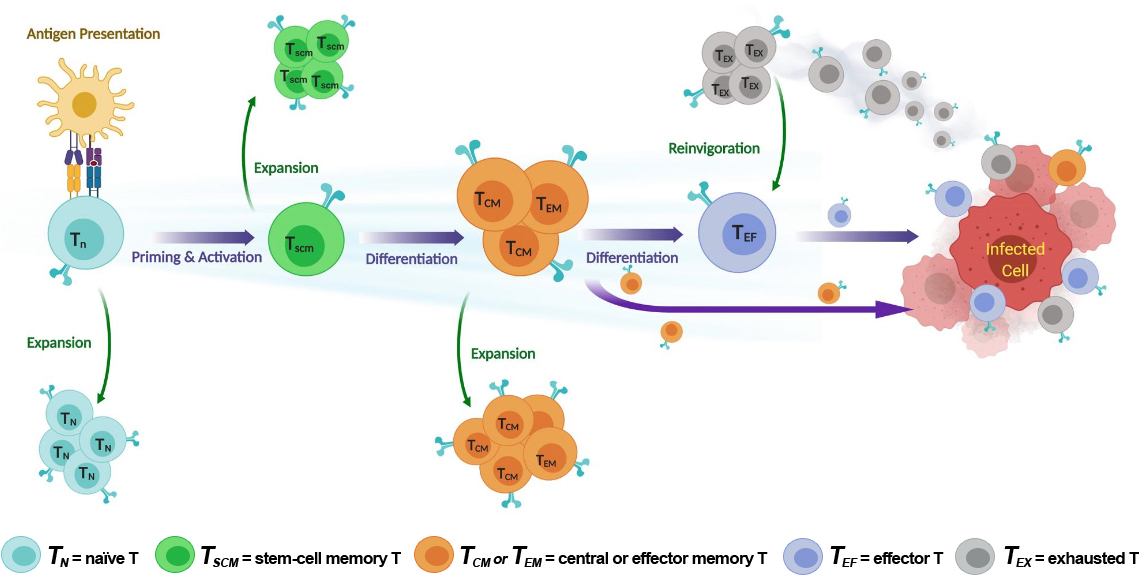
1. Maintaining homeostasis
- Supports thymopoeisis
- Boosts Tnaive cell numbers
- Maintains stable pools of diverse resting peripheral T cells, including Tscm
2. Initial Immune Response
- Boosts Tnaive cell numbers and broadens TCR repertoire diversity
- Increases sensitivity to weaker antigens
- Synergizes with TCR signaling
3. Adaptive Immune Response
- Promotes Tmemory differentiation and turnover
- Limits T cell exhaustion
- Sustains immune response to chronic antigen stimulation
NeoImmuneTech has Initiated Clinical Trials to Evaluate the Potential Benefit of NT-I7 for Immune Reconstitution and Enhancement in Vulnerable Populations.
- In NCT04054752, NT-I7 is being evaluated for its safety, tolerability, and the enhancement of immune reconstitution and vaccine responses in elderly cancer survivors following chemotherapy.
- NT-I7 received an orphan drug designation for treatment of Idiopathic CD4+ Lymphocytopenia (ICL), a rare disorder of immune system, from the EMA in 2017 and the U.S. FDA in 2019. NIT is working closely together with NIH investigators to develop a clinical study evaluating NT-I7’s potential therapeutic effect on ICL patients.
Visit our Scientific Publication section for more information on NT-I7 for immunological disorders.
NIT is collaborating with major cancer centers and universities in the U.S. and South Korea for our clinical trials with NT-I7. To learn about our clinical trials for immunological disorders, please click Here .
Scientific Publications
NT-I7 Posters and Presentations – Clinical Studies
- Heo M, Sohn J, Lee M, Phase 1b study of GX-I7, a long-acting interleukin-7, evaluating the safety, pharmacokinetics and pharmacodynamics profiles in patients with advanced solid cancers. Poster Presented at SITC; Nov. 6th, 2019; National Harbor, MD.* (Click Here )
- Lee S, Choi D, Heo M, Hyleukin-7, a long-acting interleukin-7, increased absolute lymphocyte counts after subcutaneous and intramuscular administration in healthy subjects. Poster Presented at AACR; Mar. 30th, 2019; Atlanta, GA.* (Click Here )
NT-I7 Posters and Presentations – Preclinical and Translational Research in Oncology
- Campian, Jian Li, et al. "Effect of a novel long-acting interleukin-7 agonist, NT-I7, on survival in mouse models of glioma.” Abstract submitted for ASCO; May. 31st, 2019: e13516. (Click Here )
- Kim J, Hong S, Kim Y, "Hyleukin-7, the Fc-fused interleukin-7, generates anti-tumor activity by modulating both adaptive and innate immune cells in the tumor microenvironment.” Poster Presented at AACR; Mar. 30th, 2019; Atlanta, GA. (Click Here )
- Cooper ML, Staser KW, Niswonge J, "A Long-Acting Pharmacological Grade Interleukin-7 Molecule Logarithmically Accelerates CART Proliferation, Differentiation, and Tumor Killing.” Poster Presented at TCT-Transplant & Cellular Therapy: Feb. 20th, 2019; Orlando, FL (Click Here )
- Staser KW, Cooper ML, Choi J, "Modeling Sezary Syndrome For Immunophenotyping and Anti-Tumor Effect of Ucart and Long-Acting Interleukin-7 Combination Therapy.” Poster Presented at TCT-Transplant & Cellule Therapy: Feb. 20th, 2019; Orlando, FL (Click Here )
- O'Neal, Julie, et al. "In vivo efficacy of BCMA-iNKT-CAR is enhanced by NT-I7, a long-acting IL-7.” Oral Presentation Presented at 17th International Myeloma Workshop; Sep. 13th, 2019; Boston, MA. (Click Here )
- Cooper M, Staser K, Davenport J, "A long-acting pharmacological grade interleukin-7 molecule logarithmically accelerates UCAR-T proliferation, differentiation, and tumor killing.” Poster Presented at ASH Annual Meeting; Dec. 2nd, 2018; San Diego, CA. (Click Here )
- Staser KW, Cooper ML, Choi J, "Modeling Sezary Syndrome For Immunophenotyping and Anti-Tumor Effect of Ucart and Long-Acting Interleukin-7 Combination Therapy.” Poster Presented at ASH Annual Meeting; Dec. 2nd, 2018; San Diego, CA. (Click Here )
- Kim J, Choi D, Ji M, "Preclinical evaluation of the anti-tumor activity of Fc-fused interleukin-7 in both monotherapy and combination therapy.” Poster Presented at AACR 2018; Apr. 14th, 2018; Chicago, IL. (Click Here )
Scientific Publications
- Lee, Sang Won, et al. "hIL-7-hyFc, a long-acting IL-7, increased absolute lymphocyte count in healthy subjects.” Clinical and Translational Science (2020). [Epub ahead of print] https://doi.org/10.1111/cts.12800
- Choi, Young Woo, et al. "Intravaginal administration of Fc-fused IL7 suppresses the cervicovaginal tumor by recruiting HPV DNA vaccine-induced CD8 T cells." Clinical Cancer Research 22.23 (2016): 5898-5908. https://doi.org/10.1158/1078-0432.CCR-16-0423
- Lim, Jun Yeul, et al. "Biophysical stability of hyFc fusion protein with regards to buffers and various excipients." International journal of biological macromolecules 86 (2016): 622-629. https://doi.org/10.1016/j.ijbiomac.2016.02.006
- Kang, Moon Cheol, et al. "Intranasal introduction of Fc-fused interleukin-7 provides long-lasting prophylaxis against lethal influenza virus infection." Journal of virology 90.5 (2016): 2273-2284. https://doi.org/10.1128/jvi.02768-15
- Seo, Yong Bok, et al. "Crucial roles of interleukin-7 in the development of T follicular helper cells and in the induction of humoral immunity." Journal of virology 88.16 (2014): 8998-9009. https://doi.org/10.1128/JVI.00534-14
- Ahn, So-Shin, et al. "Nonlytic Fc-fused NT-I7 synergizes with Mtb32 DNA vaccine to enhance antigen-specific T cell responses in a therapeutic model of tuberculosis." Vaccine 31.27 (2013): 2884-2890. https://doi.org/10.1016/j.vaccine.2013.04.029
- Nam, Hyo Jung, et al. "Marked enhancement of antigen‐specific T‐cell responses by IL‐7‐fused nonlytic, but not lytic, Fc as a genetic adjuvant." European journal of immunology 40.2 (2010): 351-358. https://doi.org/10.1002/eji.200939271